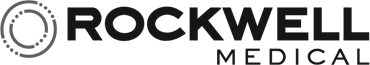Our Medical Device Projects
Gilero is a leading company in the field of medical device development and manufacturing. Gilero works with some of the most trusted brands in the world to design innovative medical devices, drug delivery systems, and combination products that improve the quality of life for patients and healthcare providers. Gilero has extensive experience in various therapeutic areas, such as cardiology, neurology, oncology, and diabetes. Gilero also offers regulatory, quality, and engineering services to support its clients throughout the product lifecycle.
A Safe, New Closed System Transfer Device
Yukon Medical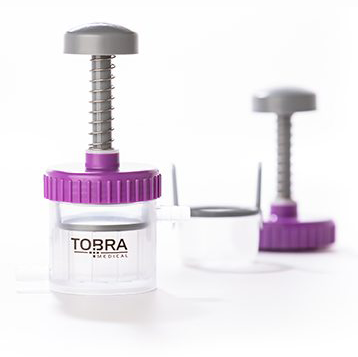
Bone Dust Collector
Tobra Medical
Buffered Anesthetic Delivery System
Anutra Medical
Capnography Mask
Salter Labs
Cell Expansion System

Connected/Electric Syringe Accessory
Assure Technologies
Empty IV Bag Solution - SmartSite Bag (with IFU)
Becton Dickinson
ENT Electromechanical Device

Hemostasis Device
Forge Medical
Home Hemodialysis Machine
LED-UV Light Disinfection System
Luer Lock Syringe

Manufacturing Transfer Project

Minimally Invasive Surgical Suturing Device
Cypris Medical
Nasal Spray

Needle-free Vial Access Device
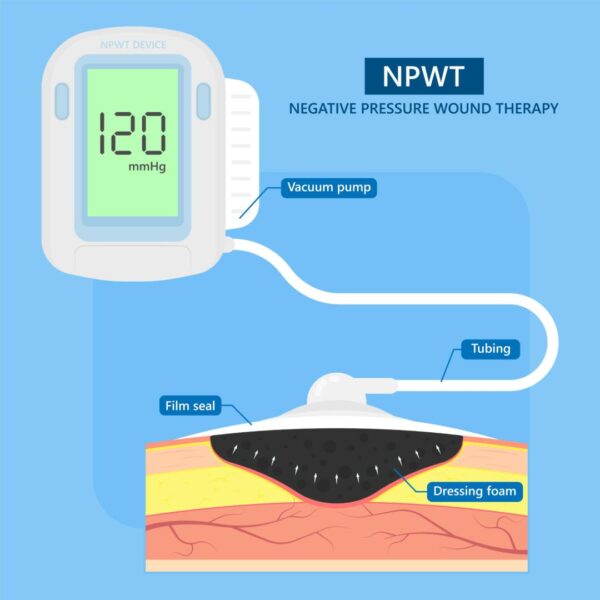
Negative Pressure Wound Therapy (NPWT) Device

Ophthalmic Drug Delivery System
Phantom Heart Pump
Myocardial Solutions
Preservative Free Eye-Dropper

Protective Face Shield
Gilero
Speed-Torque: An Elevated Torque Device
Vascugenix
Sterile Topical Anesthetic Spray
623 Medical
Syringe Infusion System
Topical Applicator for Antiseptic
Otsuka Pharmaceutical
Vented and Closed Needle-free Valve Vial Adapter